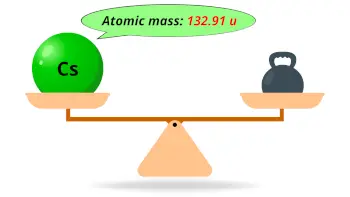
Note that this frequency occurs when the Cesium-133 atom is in isolation at absolute zero temperature in reality corrections for this are necessary. Upon observing exactly 9,192,631,770 periods (or cycles) of this electromagnetic wave, a second is said to have passed. In simpler terms, radiant energy, or light, in the form of microwaves is emitted at a very precise frequency from this particular atomic transition. The second is the duration of 9,192,631,770 periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the caesium 133 atom. Nowadays, Cesium-133 is used as the definition for the second due to the reliable frequency of microwave it emits. Aside from the fact that there are different definitions for what a "day" is, using the Earth's rotation as a definition for time is not practical due to irregularities in the frequency of its rotation. Our most common unit of time, the second, was originally defined based on the Earth's rotation the Earth rotates around its axis once per day, with the second being defined as 1/86,400 of a (mean solar) day. Cesium is the standard for atomic clocks, and actually provides the definition for the second! Some of its properties are listed below: Atomic weight Ĭesium is the 55 th element on the periodic table of elements. Cesium, with atomic number 55 and atomic weight of 132.905. Lide, (ed.), CRC Handbook of Chemistry and Physics 1999-2000 : A Ready-Reference Book of Chemical and Physical Data (CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, Florida, USA, 79th edition, 1998. I am grateful to Professor Pekka Pyykkö (University of Helsinki, Finland) who provided the nuclear quadrupole moment data in this and the following two references. Where given, data for certain radioactive nuclei are from this reference. Mason in Multinuclear NMR, Plenum Press, New York, USA, 1987. I am grateful to Professor Robin Harris (University of Durham, UK) who provided much of the NMR data, which are copyright 1996 IUPAC, adapted from his contribution contained within this reference. 5, John Wiley & Sons, Chichester, UK, 1996. Harris in Encyclopedia of Nuclear Magnetic Resonance, D.M. Magnetogyric ratio, γ (10 7 rad T ‑1 s -1) Table of NMR-active nucleus propeties of caesium Kuchitsu in Quantities, Units and Symbols in Physical Chemistry, Blackwell Scientific Publications, Oxford, UK, 1988. 
Masses, nuclear spins, and magnetic moments: I.For further information about radioisotopes see Jonghwa Chang's (Korea Atomic Energy Research Institute) Table of the Nuclides.Naturally occurring isotope abundances: Commission on Atomic Weights and Isotopic Abundances report for the International Union of Pure and Applied Chemistry in Isotopic Compositions of the Elements 1989, Pure and Applied Chemistry, 1998, 70, 217.This table gives information about some radiosotopes of caesium, their masses, their half-lives, their modes of decay, their nuclear spins, and their nuclear magnetic moments. Further data for naturally occuring isotopes of caesium are listed above.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
